Concentrated Citrus Poliphenols Extracts

Citrus fruits contain pretty high concentration of Flavanone glycosides Hesperidin and Naringin, plus smaller amount of many others flavonoids.
In well ripened fruits, flavonoids are mainly located in flavedo and in cores. Hesperidin is contained in sweet and bitter oranges, in lemons, in mandarins, in leaves, in twigs, nark and blossoms. Its biological purpose in citrus has not been completely identified, but taxonomic significance is evident. An orange contains, in average, 1,0 g of Hesperidin; processing wastes contain about 400 g/ton. As already mentioned, small size, immature fruits are very rich in Hesperidin (about 10 % by weight on dry matters) and content decreases at full maturity (< 0,5 % by weight on dry matters).
Traditional system for Hesperidin extraction involve peel treatment at pH 11,0 with calcium hydroxide. Use of lime rather than NaOH is necessary to allow release of bound water and prevent solubilization and hydrolysis of pectin avoiding large viscosity increases and slurry handling problems. After extraction, the alkaline slurry is clarified by pressing and filtration and the solution is adjusted to pH 4,0 - 4,5 . This solution is held for 24 - 48 hours, allowing Hesperidin to crystallize, after which it is recovered by filtration. The product, crude Hesperidin (about 40 %) is dried and can be be further purified. Residue filtrate can be concentrated to increase concentration and is held for a second crystallization to improve recovery.
Hesperidin has beneficial effects on permeaility and fragility of capillaries and can be transformed in sweeteners and azoic pigments.
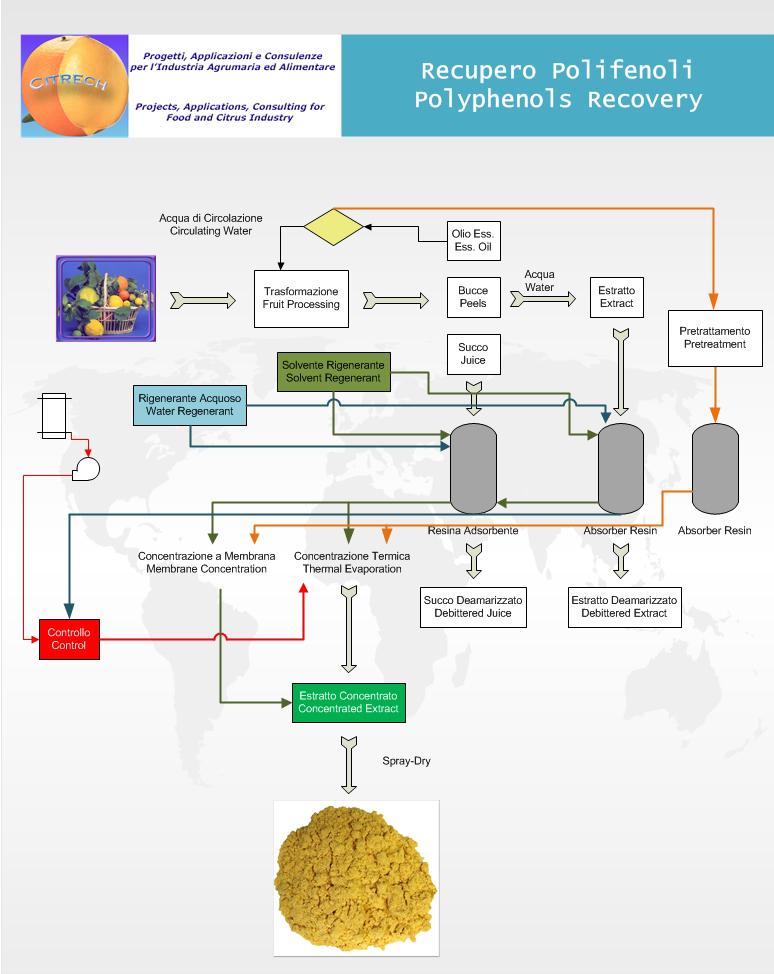
CITRECH has know-how to offer to interested citrus processors new technologies that afford to recover polyphenols in concentrated form.
As usual for CITRECH, technology is easily coupled into existing production lines; it is innovative and does not require any peels treatment like the traditional one.
With CITRECH technology You will be able to recover flavonoids both from endocarp and mesocarp (fruit's inside) and/or from esocarp (peel's outside) obtaining extracts which differs for chemical composition teflecting starting material. You'll will be entitled to choose if using solvents or not and products obtained can be thermally or membrane concentrated.
Please, see below for different possibilities: